BACKGROUND: Unprecedented reductions in multiple myeloma (MM) tumor burden after treatment with B cell maturation antigen (BCMA) directed bispecific T-cell engagers, or chimeric antigen receptor modified T-cells, have translated into improved progression free and overall survival; however, virtually all patients are predicted to ultimately relapse. The mechanisms of resistance to BCMA targeting agents are currently being explored, but presumably reflect persistence of MM cell subpopulations that either lack target antigen or otherwise evade immune-mediated tumor cell killing. Antigen targeted delivery of radiation to tumor cells leverages a unique effector mechanism that capitalizes on the exquisite sensitivity of malignant plasma cells to radiation. The α-emitter astatine-211 ( 211At) has particular promise as it deposits a very large amount of energy (~100 keV/μm) within a few cell diameters (50-90 μm) resulting in irreparable double-stranded DNA breaks. The high energy cell killing mediated by 211At is agnostic to MM cell heterogeneity, facilitates bystander delivery of radiation to target antigen negative clones, and limits radiation exposure to normal cells. We hypothesized that 211At targeting BCMA may be uniquely suited to eliminate residual MM cells.
METHODS: We conjugated a human IgG1 anti-BCMA mAb and an isotype matched nonbinding control mAb (ofatumumab), with the amine-reactive labeling agent B10-NCS to enable 211At radiolabeling. In biodistribution studies of 211At-BCMA-B10 and 211At-ofatumumab-B10 using NOD.Cg-Rag1 tm1MomIl2rg tm1Wjl/SzJ (NRG) mice bearing flank MM tumor cell xenografts (MM1R or NCI-H929; n=5 mice/group), we demonstrated tumor-specific uptake of BCMA-B10 relative to control mAb and identified the optimal dose of 211At-mAb-B10 (210μg) for assessing therapeutic efficacy. In subsequent therapy studies, NRG mice (n=7-10/group) received 0.3-0.4e6 NCI-H929 luc tumor cells by tail vein injection 6-7 days prior to administration of 210 µg of 211At-BCMA-B10 or 211At-ofatumumab-B10 or no therapy (non-treatment control). Animal body weight was monitored serially, and disease was tracked using in vivo bioluminescence imaging (IVIS Spectrum) performed at 8 or more timepoints over 150 days.
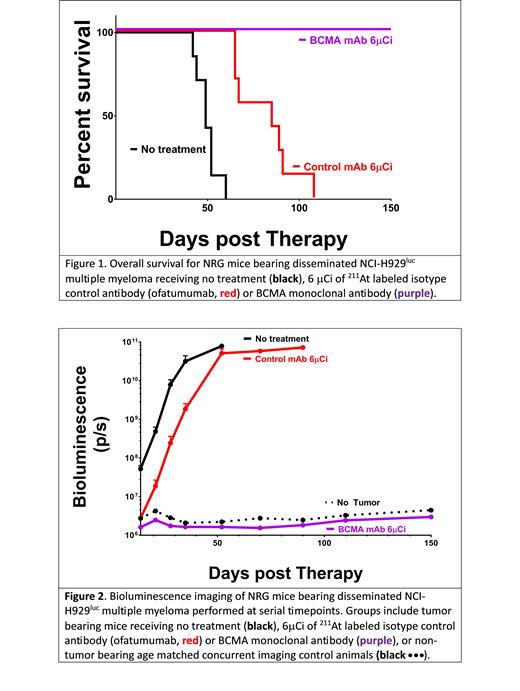
RESULTS: Disease elimination was defined as survival with no measurable disease by IVIS at 150 days following treatment. All mice receiving 6 or 8 µCi of 211At-BCMA-B10 (n=36) were cured (100%) after a single infusion of 211At-BCMA-B10. Mice in the untreated control groups uniformly experienced exponential tumor growth. At the lowest 211At dose (6 µCi), all BCMA targeted mice survived for more than 150 days while no untreated control mice survived beyond day 60 (median duration of survival 49 days [range 42-60 days]). Mice receiving 211At-ofatumumab had expected attenuation in tumor growth relative to untreated controls (presumably a consequence of non-specific radiation exposure); with a median duration of survival of 85 days; no mice survived beyond 108 days (range 65-108 days) [Figure 1, Figure 2]. No significant toxicity was observed in the 211At-BCMA-B10 treated groups. Among mice receiving 8 μCi of 211At-BCMA-B10 median animal body weight on day 14 post-treatment was 106% of baseline (range 97%-109%). Among mice receiving 6 μCi of 211At-BCMA-B10 the median body weight on day 14 was 107% of baseline (range 100% to 111%) and on day 150 the median weight was 123% of baseline (range 116% to 126%).
CONCLUSIONS: Tumor responses are encouraging with 100% of mice cured of MM after receiving low doses of BCMA targeted 211At. While we have previously demonstrated that 211At-CD38 could eliminate MM in a similar model system, in that setting fewer than 50% of mice bearing the same NCI-H929 Luc tumors, and treated with 8 μCi of 211At, survived to day 150 [O'Steen S. Blood, 2019]. In contrast, here we show that 100% of mice are cured after receiving 6 μCi of 211At-BCMA-B10. These findings support further evaluation of BCMA targeted 211At in other tumor model systems and in clinical trials aimed at eliminating minimal residual disease.
Disclosures
Orozco:Actinium Pharmaceuticals: Other: Site PI for clinical trials sponsored by Actinium, Research Funding. Walter:Amgen, Aptevo, Celgene, Janssen, Jazz, MacroGenics, Pfizer: Research Funding; ImmunoGen, Jura: Consultancy, Research Funding; Abbvie, Adicet, Amphivena, BerGenBio, Bristol Myers Squibb, GlaxoSmithKline, Orum: Consultancy. Till:BMS/Juno Therapeutics: Research Funding; Mustang Bio: Consultancy, Patents & Royalties, Research Funding; Proteios Technology: Consultancy, Current holder of stock options in a privately-held company. Hill:Applied Molecular Transport: Research Funding; iTeos Therapeutics: Research Funding; Neoleukin Therapeutics: Consultancy; iTeos Therapeutics: Consultancy; Laevoroc Oncology: Research Funding; Heat Biologics: Research Funding; Syndax Pharmaceuticals: Research Funding; Genentech: Research Funding; NapaJen Pharma: Consultancy; Commonwealth Serum Laboratories: Consultancy; Cynata Therapeutics: Consultancy; Compass Therapeutics: Research Funding; Serplus Technology: Research Funding; Generon Corporation: Consultancy. Sandmaier:Actinium Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees. Green:Ensoma: Consultancy; Janssen Biotech: Consultancy, Research Funding; SpringWorks Therapeutics: Research Funding; Celgene: Consultancy; GlaxoSmithKline: Membership on an entity's Board of Directors or advisory committees; Seattle Genetics: Consultancy, Research Funding; Juno Therapeutics A BMS Company: Patents & Royalties, Research Funding; Sanofi: Research Funding; Cellectar Biosciences: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal